World Leprosy Day 2019
World Leprosy Day – 27th Jan 2019

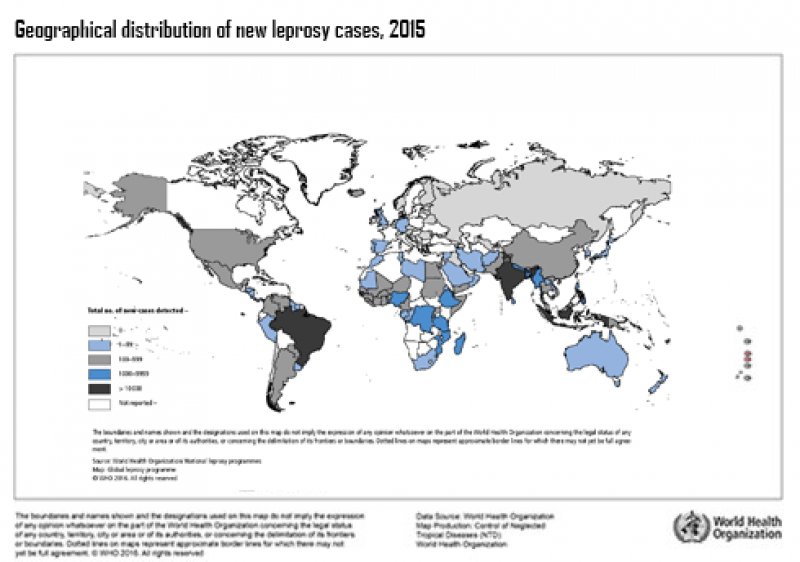
World Leprosy Day is observed annually on the last Sunday of January around the world* to raise public awareness of leprosy, a disease that is popularly believed to have been eliminated but in reality still affects around 200,000 new people every year. According to the WHO, leprosy is still prevalent in 14 countries, primarily affecting those living in poverty. We still have many knowledge gaps about the biology of Mycobacterium leprae, the bacterium that causes the disease - for example, scientists are unsure exactly how a person acquires Mycobacterium leprae - and we do not yet have a vaccine that prevents this debilitating disease.
This year World Leprosy Day will be observed on the 27th of January. To mark the occasion, we spoke to a couple of VALIDATE members who are working on vaccine development for leprosy to find out more about this disease, their research, and the challenges of vaccine development.
*In India, World Leprosy Day is held on the 30th of January every year, as this is the anniversary of the death of Mahatma Ghandi who helped those affected by leprosy.

Dr Hua Wang – Postdoctoral researcher, Francis Crick Institute
What is leprosy?
Leprosy is a chronic disease caused by the bacterium called Mycobacterium leprae that affects the peripheral nerves, with tropism for Schwann cells, resulting in loss of sensation and paralysis. Leprosy is also a profoundly social disease, with those infected suffering from not only biological ailment but also social stigma. Indeed, leprosy patients are often seen to have moral shortcomings and the disease is commonly understood to be the consequence of a divine intervention. This can be traced back to leprosy-like diseases that have been described in ancient texts as early as circa 1400 B.C. in India, mentioned as kushtha in the Vedas, and in the translations of the Bible from Hebrew to Greek, circa 300 B.C., where the word leprosy originated from the Hebrew word tsaraath, translated in Greek as lepra.
Historically, leprosy was the first disease to be linked to a microorganism, marking a scientific paradigm shift in the late half of the nineteenth century when infectious diseases were understood to be miasmatic. Prior to this discovery, commonly referred to as the germ theory of disease, leprosy was considered to be hereditary and was prevalent in Europe, with the highest prevalence in Norway, which is where most leprosy research was taking place. The idea that leprosy was hereditary was challenged by a young physician named Gerhard Henrik Armauer Hansen, when he observed little rods (bacteria) in unstained and stained specimens in 1871 but later published in 1874 titled ‘Spedalskhedeus Årsager’ (Causes of Leprosy) in Norsk Magazin for Laegeviteuskapen, to suggest that leprosy is of bacterial origin.

Leprosy sufferer, Munger Leper Colony India
How does one contract leprosy and what are the symptoms? Can you treat leprosy?
How a person acquires Mycobacterium leprae and develops the disease remains uncertain. There are studies that suggest Mycobacterium leprae is spread via contact, respiratory route, or even insects, however, the exact mechanism of transmission is not known.
The symptoms for leprosy include skin lesion (reddish or copper-coloured) with definite sensory loss, with and without thickened nerves. Damaged nerves can lead to numbness and muscle weakness and, if left untreated, leprosy can result in paralysis and even blindness. Diagnosis of leprosy is based on the symptoms and confirmed by skin smear and using a stain to detect the Mycobacterium leprae under the microscope. There is a lot of uncertainty around the pathogenesis of leprosy. Recent laboratory studies from Masaki et al. (Cell 2013) and Madigan et al. (Cell 2018) suggest that Mycobacterium leprae can reprogram the adult Schwann cells to progenitor/stem-like cells of mesenchymal traits for bacterial dissemination and that it is the reactive nitrogen species from the macrophage as an immune response to Mycobacterium leprae that damages the peripheral nerves.
Leprosy is curable. The WHO recommendation is a multidrug therapy (MDT) for leprosy, as treatment with one drug will result in development of drug resistance, with the combination of drugs depending on the classification of the disease.
Why are you interested in leprosy research?
I think leprosy is an important disease to study for both biological and social reasons. Biologically, there is very little knowledge about the bacterium Mycobacterium leprae. For example, the Mycobacterium leprae genome is half pseudogenes (non-functional genes) and half functional genes (coined as an example of a minimal set of genes for survival and pathogenesis). The role and function of pseudogenes has been a scientific mystery ever since their discovery. However, recent studies suggest that pseudogenes do have biological functions (Pink et al., RNA 2011). Moreover, a recent report by Singh et al. (PNAS 2015) shows that leprosy is not exclusively caused by Mycobacterium leprae. These work raises the fundamental questions about pseudogenes and bacterial evolution in relation to human migration that the study of Mycobacterium leprae offers unique potential to explore. From a social perspective, there is also a lot of work left to be done with regards to leprosy. Leprosy is a highly stigmatised disease. In bridging together both the biological and social gaps in knowledge, there is great potential for unconventional thinking and transdisciplinary approach to ask questions that could offer new ideas about bacteria and bacteria-human relationship.
In a way, my interest in leprosy research is a nod to a quote by Georges Canguilhem, "in calling such a theory a dogma we do not mean at all to disparage it, but rather to stress its scope and repercussions." I think leprosy research offers a restart button, an opportunity to explore our current understanding of bacterial diseases with an unconditional peripheral view.
What is your research about?
My research seeks to understand bacteria on a basic or fundamental level, from the perspective of bacteria itself. More specifically, I am interested in the function of bacterial pigments from Mycobacterium leprae and other pathogenic bacteria. At the moment, there is very little knowledge about the biological fate of bacterial pigments. Thus, studying bacterial pigments opens up space to understand their physiological properties within the cell and their physiological implications in humans (e.g. immune response and manifestation of the disease). Moreover, at the moment, even the basic laboratory method of cultivating Mycobacterium leprae is not possible or practical because Mycobacterium leprae cannot be cultivated using artificial medium. The current method of cultivating the bacteria resorts to using animal models, such as mouse foot-pad that was developed by Charles Shepard in 1959, or nine-band armadillo that was demonstrated by Waldemar Kirchheimer and Eleanor Storrs in 1968. These methods have many limitations and concerns. A goal of my research is in line with the principles of 3Rs (Replacement, Reduction and Refinement) to reduce or eliminate the use of animals to culture Mycobacterium leprae, by establishing and developing an experimental model system to specifically study the biological properties of pigments produced by Mycobacterium leprae and also using the same system to study how Mycobacterium leprae infects humans, how Mycobacterium leprae evades the immune response, and how Mycobacterium leprae causes neuropathy
How will your research impact the development of a leprosy vaccine?
Mycobacterium leprae biology remains largely underexplored. My research aims draw upon the unique nature of Mycobacterium leprae in order to establish a new experimental model for microbiological research in general. This method in combination with new biological tools and technologies at our disposable, I propose, could be used to identify and study unique surface proteins or molecules from Mycobacterium leprae that illicit host immune response for vaccine development.

Dr Rachel Tanner – Postdoctoral researcher and VALIDATE Fellow, University of Oxford
Where are we with a vaccine for leprosy and why is one important?
There is currently no vaccine specifically for leprosy. BCG was developed as a vaccine against tuberculosis (TB), and it also affords some protection against leprosy because Mycobacterium tuberculosis and Mycobacterium leprae are highly related. However, BCG is protective against leprosy to very varying degrees and only in some populations. Even where protection is observed, it drops off over time. Leprosy remains endemic in some countries where BCG vaccination is widespread, so we clearly need to improve on BCG.
There are few leprosy vaccines in development, although three are currently in Phase 3 clinical trials which means they have shown promise and are now being tested on a large scale in many hundreds of subjects. One of these is Mycobacterium indicus pranii (MIP), which has shown the highest efficacy ever reported against leprosy (68%) for a vaccine other than BCG in one study of household contacts.
A successful leprosy vaccine would be a critical step in controlling the epidemic by preventing disease and transmission. Although other strategies include the development of new drugs and reducing antibiotic resistance, prevention is always better than cure.
What are the challenges with developing a leprosy vaccine?
Mycobacterium leprae is an intracellular pathogen so it can effectively ‘hide’ from the immune system. It is also notoriously difficult to work with, as it cannot be cultured in artificial medium (so making large amounts of vaccines based on Mycobacterium leprae itself is challenging), and the only animal models available are nine-banded armadillos and mouse footpads. As is the case for TB as well as leprosy, we don’t understand which parts of the human immune system are important in protection from these infections. This makes it difficult to design a vaccine because we don’t know what it needs to do! It’s also unclear why BCG is effective in some populations and not others, and we don’t want a new vaccine to be subject to the same pitfalls.
Mycobacterium leprae
What is your research about?
I am studying the antibody response to BCG in humans, cattle and non-human primates, and trying to identify the bacterial proteins recognised by these antibodies. I can then relate the responses to how protected the same animals were from previous TB or bovine TB infection experiments to select antibodies that might play a role in protection. I am particularly interested in recognised proteins that are present across Mycobacterium leprae, Mycobacterium tuberculosis and Mycobacterium bovis. I will use these findings to design new vaccines with the aim of inducing much higher levels of the antibodies and then test whether these are protective in mice.
What do you want to achieve with your research?
Because of the intracellular nature of mycobacteria, most vaccine research to date has focussed on the cellular response, but there is increasing evidence that antibodies may also play a role in protection from TB. Furthermore, we now recognise that the cellular and humoral immune responses interact, so it’s possible that a successful vaccine would harness both types of response. Although it’s unlikely that a successful vaccine will be based on antibodies alone, I hope that in the future the proteins I identify can be combined with those that generate a strong cellular response. Such a vaccine may be more effective against leprosy, TB and bovine TB.
Published: 25 January 2019


