Melioidosis
About Melioidosis
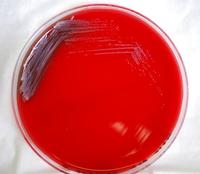
Burkholderia pseudomallei on blood agar plate (credit: CDC)
Melioidosis is a disease caused by a bacterium, Burkholderia pseudomallei, and is found across tropical and semi-tropical regions worldwide. It is estimated that 165,000 people develop melioidosis annually with 89,000 of these cases being fatal, making melioidosis a global health concern.
Treatment of melioidosis is difficult with 43% of people with the disease dying in NE Thailand [1] and 14% in N Australia [2]. The occurrence of melioidosis in the population is associated with some risk factors such as diabetes and generation of a vaccine and administration to at risk personnel has been demonstrated to be potentially cost effective [3].
Where is melioidosis found?
Melioidosis occurs in tropical and semi-tropical regions of the world, especially in rice farming regions. B. pseudomallei can survive in the environment for long periods of time, present in the soil and water in endemic areas. The melioidosis.info webpage provides maps showing distribution of the disease, cases diagnosed and mortality.
How is melioidosis contracted?
Infection is acquired through skin inoculation, contamination of wounds, inhalation or ingestion.
What are the symptoms of melioidosis?
Diagnosis of melioidosis is difficult with symptoms varying depending on route and stage of infection. Often initial symptoms resemble many other bacterial infections with fever, and severe septicaemia (blood poisoning) can develop.
Is melioidosis curable?
Melioidosis is curable but B. pseudomallei is naturally drug-resistant making treatment choices limited and sometimes ineffective.
Treatment takes about 4 months with 10-14 days intravenous therapy followed by 12 weeks of oral therapy. This is very costly, and can have unpleasant side effects. In addition there are a percentage of individuals who will enter a latent (dormant) infection even after the intensive antibiotic therapy. These people may go onto have a resurgence of infection weeks, months or even years later. This means developing a vaccine that could prevent melioidosis infection is an important global public health goal.
Vaccines against melioidosis
There is no vaccine for melioidosis. There are vaccine research programmes currently focusing on inactivated bacteria, live attenuated (altered) bacteria and subunits (parts of the bacteria).
How is VALIDATE helping?
At the VALIDATE Network, by bringing together melioidosis researchers from around the world we hope to speed up progress towards a vaccine against this deadly disease.
Further Information (also see our Publications page)
Chakravorty A et al 2019. Melioidosis: an updated review. Aust J Gen Pract 48(5):327-332
Limmathurotsakul et al 2016. Predicted global distribution of Burkholderia pseudomallei and burden of melioidosis. Nature Microbiology 1, Article number: 15008.
Melioidosis. Nat Rev Dis Primers 4, 17108 (2018)
References
1. Limmathurotsakul D, et al 2010. Increasing incidence of human melioidosis in Northeast Thailand. Am J Trop Med Hyg 82: 1113–1117.
2. Currie BJ, Ward L, Cheng AC 2010. The epidemiology and clinical spectrum of melioidosis: 540 cases from the 20 year Darwin prospective study. PLoS Negl Trop Dis 4: e900.
3. Peacock et al 2012. Melioidosis Vaccines: A Systematic Review and Appraisal of the Potential to Exploit Biodefense Vaccines for Public Health Purposes. PLOS Negl Trop Dis 6: e1488.
Burkholderia pseudomallei is a pathogen that infects humans causing melioidosis, a non-transmissible disease resulting in high mortality and morbidity.
Evidence is evolving to suggest that B. pseudomallei is present in many tropical and sub-tropical regions of the world [1,2]. Identification and diagnosis are problematic due to non-specific symptoms of infection and inaccurate identification because of cross reactivity with closely related environmental bacteria. Treatment of infection can be ineffective due to inherent antibiotic resistance of the pathogen resulting in lengthy treatment regimes with high rates of failure. B. pseudomallei is designated as a select agent by the centre for disease control because of its potential as a bioweapon and its ability to cause lethal disease in humans and animals.
The challenges of developing a Melioidosis vaccine
Developing an effective B. pseudomallei vaccine is a considerable challenge. There are several different types of vaccine in the research phase, demonstrating varying levels of efficacy in a number of animal models. Progress in the B. pseudomallei vaccine field of research is impeded by the lack of an agreed animal vaccine challenge model. This results in generation of data from a number of different laboratories that cannot be directly compared. There is a requirement for both a public health and military vaccine. In the public health case the vaccine would need to protect humans against infection from ingestion, aerosolisation or subcutaneous injection of the pathogen, whereas the most likely route of exposure in a military scenario would be by aerosolisation.
In order to progress a potential B. pseudomallei vaccine it may be that the ‘animal rule’ to obtain licensure would be used. This requires that animal efficacy data is compiled along with human safety data to progress a vaccine to licensure. This is because it may be difficult to generate sufficient data for a phase 3 trial in the disparate at risk human population.
Further challenges include:
Lack of immune correlates
For some vaccines, the level and particular kind of immune response that is needed for the vaccine to work – i.e. to stop someone getting disease, is known. For melioidosis, we do not know what kind of immune response is needed, let alone what level of response is required.
Uncertain predictive value of animal models
It is not possible to test a vaccine in humans without testing it first in animals. However, we do not know which, if any, of the animal models to assess B. pseudomallei vaccines best predicts efficacy in humans. This means we can only determine if a vaccine will evoke a potentially appropriate immune response in humans by immunogenicity testing.
Difficulty of working with B. pseudomallei in the lab
B. pseudomallei is a highly infectious pathogen, which means we can only safely work with it in the lab using special containment measures (called Category or BSL 3). This limits the number of laboratories that can work on B. pseudomallei.
How VALIDATE is helping
By bringing together researchers working on different (but similar) pathogens, discoveries in one field can be more quickly taken advantage of in research against another pathogen. Bringing together researchers from different disciplines and institutes in new collaborations means knowledge can be exchanged and new research ideas for the field can be generated and investigated. Bringing new researchers into this field, and progressing the careers of early career researchers, will aid with new ideas and the continuation of the field into the future.
Further reading (find more on our Publications page)
Luangasanatip N et al 2019. The global impact and cost-effectiveness of a melioidosis vaccine. BMC Medicine
Rongkard P et al 2020. Human Immune Responses to Melioidosis and Cross-Reactivity to Low-Virulence Burkholderia Species, Thailand. Emerging Infectious Diseases 26(3):463-471.
The ‘Melioidosis.info’ webpages also ave a lot of useful information.
References
1. Limmathurotsakul D et al. Predicted global distribution of Burkholderia pseudomallei and burden of melioidosis. Nat Microbiol 2016;1:15008.
2. Currie BJ, et al. The global distribution of Burkholderia pseudomallei and melioidosis: an update. Trans R Soc Trop Med Hyg 2008;102 (Suppl 1):S1–4.

Establishment of a functional assay panel to evaluate the role of antibodies in defence against melioidosis and tuberculosis
Led by Dr Panjaporn Chaichana (MORU, Thailand), with Prof Susanna Dunachie (University of Oxford, UK), and Prof Helen Fletcher (LSHTM, UK)
Read more here

In vivo protection studies of chimeric Burkholderia pseudomallei antigens presenting multiple epitopes on protein scaffolds and outer membrane vesicles
Led by Prof Gregory Bancroft (LSHTM, UK), with Asst Prof Louise Gourlay (University of Milan, Italy), Prof Martino Bolognesi (University of Milan, Italy), Prof Giorgio Colombo (University of Pavia, Italy), and Asst Prof Ganjana Lertmemongkolchai (Khon Kaen University, Thailand)
Read more here

Vaccines to target people with diabetes: characterising the pathways of immune response to M. tuberculosis and B. pseudomallei in people with diabetes compared to non-diabetics
Led by Prof Susanna Dunachie (University of Oxford, UK), with Asst Prof Jacqueline Cliff (LSHTM, UK), and Prof Gregory Bancroft (LSHTM, UK)
Read more here

Developing a mouse model of diabetes to evaluate vaccines for TB and melioidosis
Led by Dr Elena Stylianou (University of Oxford, UK), with Prof Helen McShane (University of Oxford, UK), Assoc Prof Susanna Dunachie (University of Oxford, UK), Assoc Prof Paul Brett (University of Nevada, USA), Dr Barbara Kronsteiner-Dobramysl (University of Oxford, UK) and Dr Panjaporn Chaichana (MORU, Thailand)
Read more here

Elucidating the T-cell epitopes and T-cells responses of two B. pseudomallei vaccine antigens
Led by Dr Julen Tomás Cortázar (University College Dublin, Ireland), with Prof Susanna Dunachie(University of Oxford, UK), Asst Prof Louise Gourlay (University of Milan, Italy), Prof Giorgio Colombo(University of Pavia, Italy), and Dr Siobhán McClean (University College Dublin, Ireland)
Read more here

Characterising the cellular immunity and metabolic response to Mycobacterium tuberculosis and Burkholderia pseudomallei in Indian patients for vaccine design
Led by Prof Chiranjay Mukhopadhyay (Manipal Academy of Higher Education, India), with Prof Susanna Dunachie (University of Oxford, UK) and Prof Mitali Chatterjee (IPGMER, India)
Read more here


